A Molecular Psychiatry paper by Prof. Zheng Wang's group revealed a network regulation mechanism underlying neurosurgical interventions in refractory obsessive-compulsive disorder using fMRI
Recently, an article entitled “Common and differential connectivity profiles of deep brain stimulation and capsulotomy in refractory obsessive-compulsive disorder” has been published online in Molecular Psychiatry by Prof. Zheng Wang’s research group from School of Psychological and Cognitive Sciences, IDG/McGovern Institute for Brain Research and Peking-Tsinghua Center for Life Sciences at Peking University.
Obsessive-compulsive disorder (OCD) is a severe chronic psychiatric disorder with a life-time prevalence of 2%-3%, which ranks the fourth among the most common mental disorders by World Health Organization (WHO). OCD, also as one of WHO’s ten leading causes of disability. OCD not only raises a severe threat to individual’s health but also imposes a heavy economic and social burden on families and communities. Wang lab recently combined a transgenic monkey model and clinical autistic patients to elucidate the underlying pathophysiology (J Neurosci, 2020), and developed an innovative trans-species machine learning model to improve the accuracy of diagnosis of autism and OCD (Am J Psychiatry, 2021; with commentary by van den Heuvel). In addition, Prof. Wang’s group found a divergent structural response of regional gray matter volume to selective serotonin reuptake inhibitors (EBioMedicine, 2017). However, about 30% patients are refractory to these first-line therapies such as pharmacological and cognitive-behavioral therapies.
For these treatment-resistant patients, neurosurgical intervention is considered as the “last resort”. A variety of white matter and gray matter areas within the cortico-striato-thalamo-cortical (CSTC) circuit have been chosen as therapeutic targets for neurosurgical interventions, such as capsulotomy and ventral capsule / ventral striatum deep brain stimulation (DBS). By analyzing the functional connectivity of these pathways, Prof. Wang’s team found that capsulotomy significantly decreased the FCs between ventral striatum and anterior cingulate cortex (Biol Psychiatry, 2018; commentary by Hoexter MQ). Meanwhile, capsulotomy induced morphological changes of widespread gray / white matter regions, where the atrophy of caudate was correlated with treatment response (Biol Psychiatry CNNI, 2021). Interestingly, patients with more fibers projecting from dorsolateral prefrontal cortex (PFC) to thalamus showed better treatment responses (J Neurol Neurosurg Psychiatry, 2021). However, it is not clear about common and differential connectivity profiles between the network modulations of DBS and capsulotomy. How are these profiles related with clinical improvements? Is it possible to identify treatment-specific imaging markers for pre-surgical treatment-selection and optimization of treatment response in individual patients?
To address these issues, we retrospectively assembled 215 subjects with MRI scanning and clinical data, among which 82 healthy controls and 104 OCD were locally enrolled with 27 refractory OCD undergoing capsulotomy and postoperative fMRI scanning, 15 healthy controls and 14 OCD treated by DBS. Using resting-state fMRI data, we firstly built a principal component regression (PCR) based machine learning model and identified lateral PFC highly predictive of OCD symptoms. By comparing the pre-surgical functional networks, we observed a significantly biased distribution of the network effects shared by both capsulotomy and DBS, inhibiting cortico-subcortical FCs but enhancing cortico-cortical FCs (Fig. 1). Previous work by Prof. Wang’s group (Biol Psychiatry, 2016; Cereb Cortex, 2018) has adopted a graph-based approach to evaluate the topological properties of functional network. Here, we applied the network communication strength to characterize treatment-induced profile changes of cortico-cortical and cortico-subcortical connections (Fig. 2A). Both DBS and capsulotomy downregulated cortico-subcortical communication strength but upregulated cortico-cortical communication strength. Meanwhile, DBS-induced increase of cortico-cortical communication strength at ventrolateral PFC was positively correlated to mood improvements; capsulotomy-induced increase of cortico-cortical communication strength at centrolateral PFC was positively correlated to anxiety improvements (Fig. 2B).
We next asked whether and which cortical nodes with altered communication strength could serve as candidate treatment-specific imaging markers. A linear regression model with treatment (DBS or capsulotomy) and outcome (clinical score improvements [pre- minus post-treatment]) factors was launched for the pretreatment communication strength of each cortical nodes. The results showed that pretreatment cortico-subcortical communication strength of ventrolateral PFC was differentially predictive of mood improvements and pretreatment cortico-cortical communication strength of centrolateral PFC was differentially predictive of anxiety improvements (Fig. 3C). The post hoc Pearson’s correlation analyses further showed that patients with lower pretreatment cortico-subcortical communication strength in ventrolateral PFC were more amenable to DBS, whereas lower pretreatment cortico-cortical communication strength in centrolateral PFC preferred capsulotomy. In short, this study found a pattern of focal inhibition and distant enhancement commonly shared by DBS and capsulotomy which suggests an adaptive reorganization (not a passive diffusion process) mechanism in response to neuromodulation. Future work includes in-depth development of a whole-brain network model (Neurosci Bulletin, 2018) to simulate circuit dynamics and its response to focal neuromodulations, thereby providing theoretical guidance for neurosurgical interventions.
PhD student Xiaoyu Chen from CAS Center for Excellence in Brain Science and Intelligence Technology (Institute of Neuroscience) and Dr. Zhen Wang from Shanghai Mental Health Center are co-first authors of this work, and Prof. Zheng Wang is the only corresponding author. This study received generous help from Prof. Damiaan Denys from University of Amsterdam and Prof. Valerie Voon from University of Cambridge. It is funded by Key-Area Research and Development Program of Guangdong Province, National Key R&D Program of China, Strategic Priority Research Program of Chinese Academy of Science, National Natural Science Foundation and Shanghai Municipal Science and Technology Major Project.
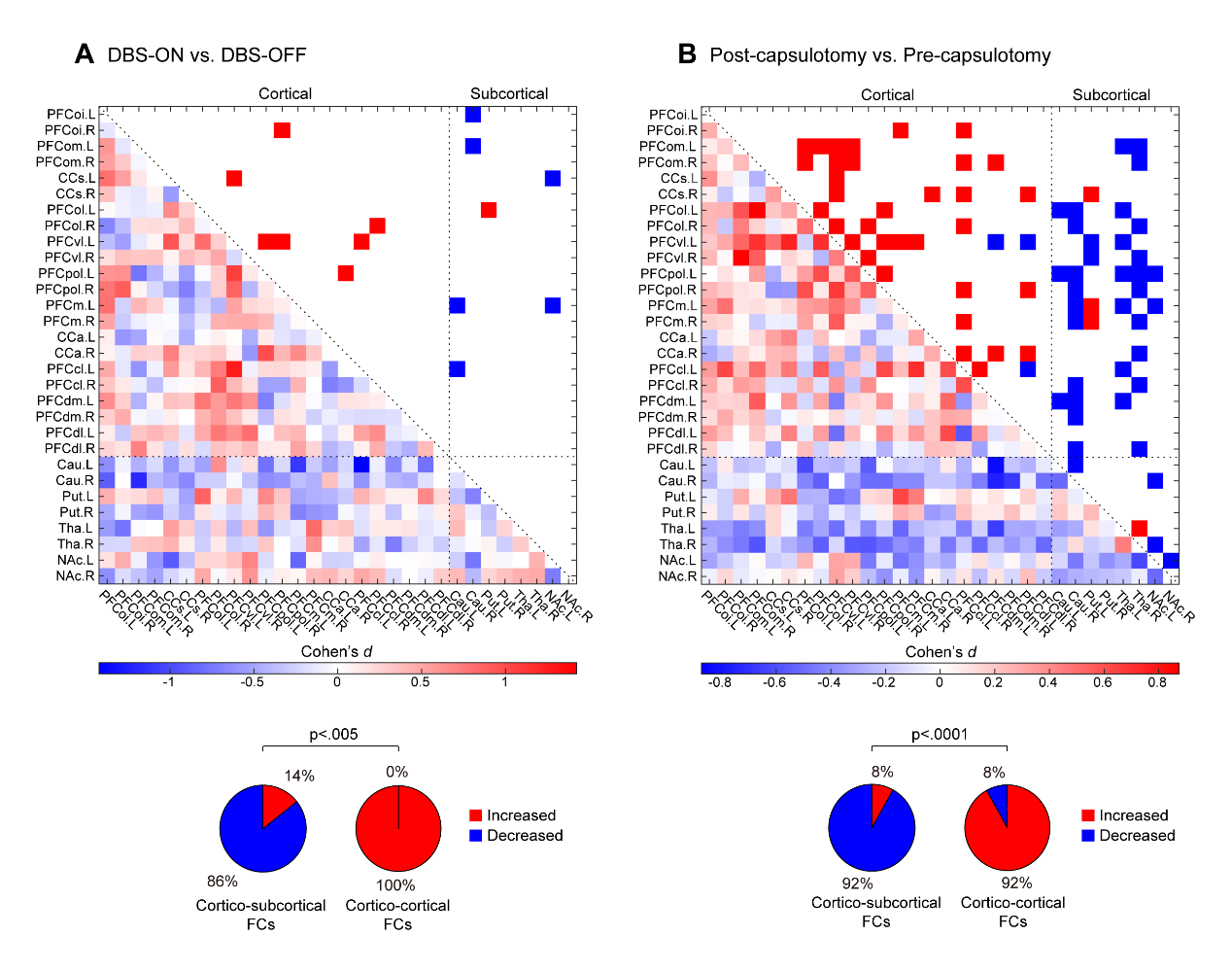
(Fig. 1:Network effects of neurosurgical interventions)
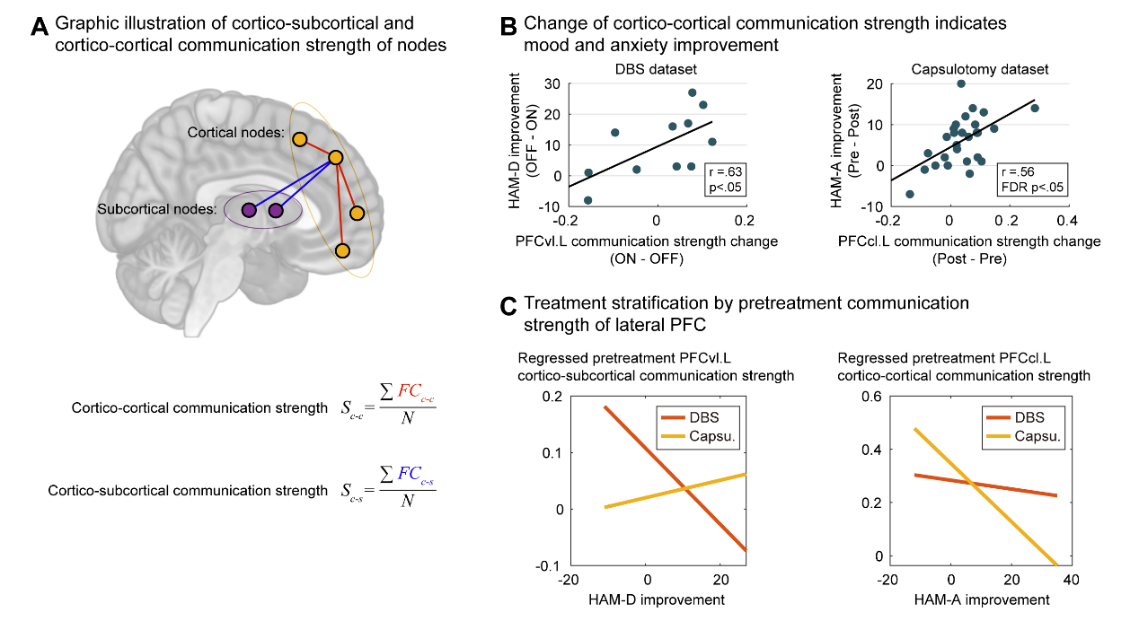
(Fig. 2:Cortico-cortical and cortico-subcortical communication strength altered by neurosurgical interventions)